Looking to save on ARAKODA?
Check for coupons on
GoodRx.
Protecting global
travelers from malaria
For individuals traveling to endemic areas, ARAKODA® (tafenoquine) tablets are indicated for the prophylaxis of malaria in patients aged 18 years and older.1
The Annual Burden of Malaria
25-30 million travelers visit malaria-endemic tropic and subtropic regions2
About 250 million cases in 85 countries and over 600,000 deaths3*
About 2,000 cases in the United States, mostly from travelers returning from sub-Saharan Africa and South Asia4
A New Standard of Care With ARAKODA
The only prophylactic therapy to provide protection against all stages of malaria1
Recommended by the CDC for global use without geographic restrictions4
Convenient weekly dosing1†
Established safety profile1
Why ARAKODA Is Different
Broad-Spectrum Activity of ARAKODA on Different Lifecycle Stages for All Species of Plasmodium Parasites in a Convenient Weekly Dosing Regimen5
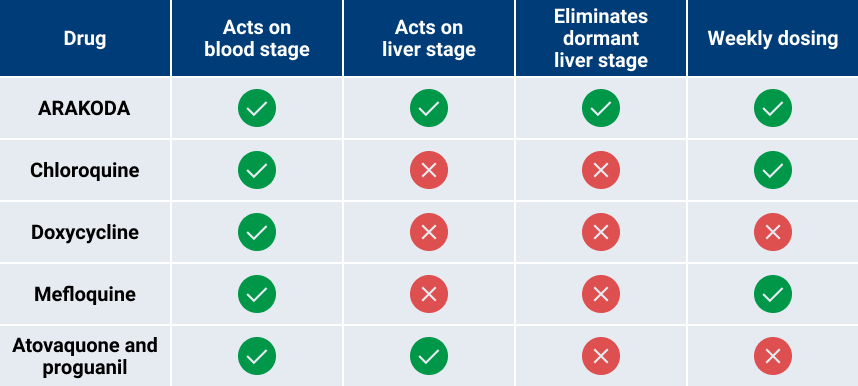
CDC = Centers for Disease Control and Prevention.
*Based on 2022 data.
†With one dose upon return from traveling. Please see Prescribing Information and dosing instructions.
View the ARAKODA Product Monograph
DOWNLOAD MONOGRAPH

ARAKODA is available through Amazon pharmacy and most major retailers.
To learn if ARAKODA is available in your region, ask your local pharmacy or visit Infuserve.com.
If you have difficulty locating or accessing ARAKODA, contact 60 Degrees Pharmaceuticals here.
Looking to save on ARAKODA?
Check for coupons on GoodRx.
Fulfilling the Promise of ARAKODA
With promising new data, 60 Degrees Pharma is seeking to evaluate the utility of tafenoquine for the following indications:
Tafenoquine Pipeline
Treatment of Babesiosis*
Tafenoquine exhibits useful activity in animal models of babesiosis and may have clinical utility in treatment of drug-resistant strains. A proof-of-concept clinical study is planned to assess the utility of ARAKODA as a treatment for human babesiosis.
Learn MoreTo learn more about our clinical trials, click here or email us at inquiries@60degreespharma.com.
Treatment and Prevention of Fungal Diseases*
In vitro and in vivo studies demonstrate that tafenoquine is effective for treatment and prevention of fungi such as Pneumocystis and yeasts at doses that are clinically relevant.6
Read More*ARAKODA has not been reviewed or approved by the U.S. Food and Drug Administration for the treatment or prevention of babesiosis or fungal diseases.
References: 1. Arakoda® [package insert]. 60 Degrees Pharmaceuticals LLC: 2021. 2. Angelo KM, Libman M, Caumes E, et al. Malaria after international travel: a GeoSentinel analysis, 2003-2016. Malar J. 2017;16(1):293. doi:10.1186/s12936-017-1936-3 3. World Health Organization. Malaria fact sheet. Updated December 4, 2023. Accessed March 4, 2025. https://www.who.int/news-room/fact-sheets/detail/malaria 4. Centers for Disease Control and Prevention. CDC Yellow Book. Malaria. Reviewed January 31, 2025. Accessed March 21, 2025. wwwnc.cdc.gov/travel/yellowbook/2024/infections-diseases/malaria 5. Data on file. 2025. 60 Degrees Pharmaceuticals LLC. 6. Dow GS, Smith BL. Tafenoquine exhibits broad spectrum antifungal activity at clinically relevant concentrations in vitro and decreases lung fungal burden in an invasive pulmonary model of Rhizopus in vivo. New Microbes New Infect. 2022;45:100964. doi:10.1016/j.nmni.2022.100964
